In a groundbreaking study, scientists from the Nara Institute of Science and Technology (NAIST) in Japan have uncovered a crucial regulator that influences the regeneration process in plants.
By investigating the intricacies of shoot regeneration in Arabidopsis, a widely studied plant species, the researchers identified and characterized the WUSCHEL-RELATED HOMEOBOX 13 (WOX13) gene as a key negative regulator, shedding new light on the mechanisms governing plant reconstruction.
Plants possess a remarkable ability to regenerate from somatic cells, which are ordinary cells not typically involved in reproduction.
This regenerative process involves the formation of a shoot apical meristem (SAM), from which lateral organs develop, playing a vital role in plant reconstruction.
At the cellular level, the formation of SAM is meticulously controlled by positive and negative regulators, such as genes and protein molecules, which either promote or hinder shoot regeneration, respectively.
However, the specific molecules involved in this intricate process and the underlying regulatory layers have remained elusive until now.
To address these pressing questions, the research team led by Momoko Ikeuchi at NAIST embarked on an in-depth investigation.
Their study, recently published in Science Advances, focused on Arabidopsis to explore the process of shoot regeneration and the role of regulatory genes.
Through their experiments, the scientists unveiled the pivotal role of the WOX13 gene and its associated protein in promoting the non-dividing function of callus cells.
They discovered that WOX13 acts as a transcriptional repressor at the RNA level, impacting the efficiency of regeneration.
“Enhancing shoot regeneration efficiency in plants has been a long-standing challenge, hampered by the lack of understanding regarding related regulatory mechanisms.
Our study addresses this knowledge gap by revealing a novel pathway for cell-fate specification,” elucidated Momoko Ikeuchi, the principal investigator of the study.
In previous studies, Ikeuchi’s team had already identified the involvement of WOX13 in tissue repair and organ adhesion following grafting.
Building on these findings, they initially examined the potential role of WOX13 in controlling shoot regeneration using a two-step tissue culture system with Arabidopsis mutants lacking functional WOX13.
Phenotypic and imaging analyses of the mutant plants revealed a significant acceleration in shoot regeneration, with regeneration occurring three days faster compared to normal plants.
Conversely, inducing WOX13 expression resulted in a slower regeneration process.
Notably, WOX13 expression levels were found to be locally reduced in the SAM of normal plants, suggesting its negative regulatory role in shoot regeneration.
To validate their findings, the researchers conducted RNA-sequencing experiments at various time points, comparing the wox13 mutants with wild-type plants.
Surprisingly, the absence of WOX13 did not significantly alter gene expression under callus-inducing conditions.
However, under shoot-inducing conditions, the alterations induced by the wox13 mutation were considerably enhanced, leading to an upregulation of shoot meristem regulator genes.
An intriguing observation was made when the researchers overexpressed WOX13 in the mutant plants.
Within 24 hours, they observed the suppression of genes associated with shoot meristem regulation.
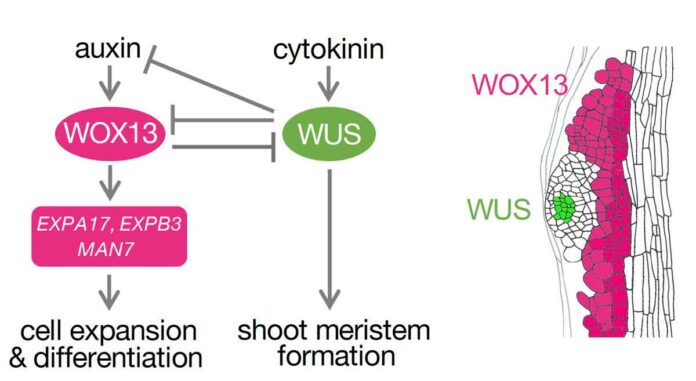
Additionally, the team found that WOX13 inhibited a subset of shoot meristem regulators while activating genes involved in cell expansion and cellular differentiation, particularly those related to cell wall modification.
These findings were further confirmed through single-cell RNA sequencing.
This study highlights a unique characteristic of WOX13 as a major regulator of shoot regeneration efficiency.
Unlike other known negative regulators, which solely prevent the transition from callus to SAM, WOX13 inhibits SAM specification by promoting the acquisition of alternative fates.
This inhibitory effect is achieved through a mutually repressive regulatory circuit involving the regulator WUS, whereby WOX13 transcriptionally inhibits WUS and other SAM regulators while inducing cell wall modifiers.
The implications of this research extend beyond the realm of scientific understanding, as it presents exciting possibilities for practical applications in agriculture and horticulture.
By knocking out the WOX13 gene, the acquisition of shoot fate can be promoted, thereby enhancing shoot regeneration efficiency.
This breakthrough has the potential to revolutionize tissue culture-mediated de novo shoot regeneration of crops, offering new avenues for crop improvement and development.
In conclusion, the study conducted by the research group at NAIST unraveled the mysteries surrounding shoot regeneration in plants.
Through the identification and characterization of WOX13 as a key negative regulator, scientists can now delve deeper into the complex regulatory networks governing plant reconstruction.
This newfound knowledge paves the way for innovative strategies to enhance shoot regeneration efficiency, with far-reaching implications for agricultural practices and crop productivity.
FAQ
The discovery of a key negative regulator, WOX13, in the process of plant regeneration opens new doors for understanding the complex mechanisms governing this remarkable ability of plants. It provides crucial insights into the cellular and genetic factors that influence shoot regeneration and offers potential applications in agriculture and horticulture.
Shoot regeneration is the process by which plants can regrow shoots, including stems, leaves, and flowers, from somatic cells. This ability is vital for plant survival and adaptation to environmental changes. Studying the factors that regulate shoot regeneration can help scientists develop strategies to enhance plant growth, improve crop yields, and facilitate tissue culture-mediated regeneration techniques.
The WOX13 gene acts as a negative regulator of shoot regeneration in plants. It promotes the non-meristematic function of callus cells, which are involved in tissue repair and regeneration. By inhibiting shoot meristem regulators and inducing cell wall modifiers, WOX13 influences the fate of pluripotent callus cells, impacting the efficiency of shoot regeneration.
The researchers used Arabidopsis, a commonly studied plant species, as a model organism. They compared normal plants with wox13 mutants and analyzed gene expression patterns using RNA-sequencing and single-cell RNA sequencing techniques. Through these experiments, they observed how the absence or overexpression of WOX13 affected shoot regeneration and identified its role in regulating specific genes involved in shoot meristem and cell wall modification.
The newfound understanding of WOX13’s role as a negative regulator of shoot regeneration opens up possibilities for enhancing tissue culture-mediated de novo shoot regeneration in crops. By manipulating the expression or function of WOX13, it may be possible to increase the efficiency of shoot regeneration, leading to improved crop yields, better disease resistance, and more effective propagation techniques in agriculture and horticulture.
The identification and characterization of WOX13 as a crucial regulator of shoot regeneration contribute to the broader understanding of the genetic and cellular mechanisms governing plant reconstruction. By unraveling the complex regulatory networks involved in plant regeneration, scientists gain insights into fundamental processes that drive growth and development in plants, potentially opening avenues for further advancements in plant biology research.
More information: Science Advances (2023). DOI: 10.1126/sciadv.adg6983. www.science.org/doi/10.1126/sciadv.adg6983