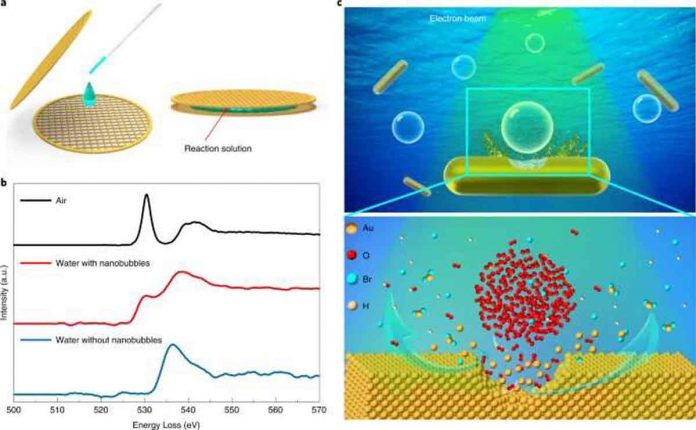
Solid-liquid-gas reactions are common in a variety of natural and industrial phenomena, including hydrogen-oxygen fuel cell reactions, heterogeneous catalysis, and metal corrosion in ambient environments. However, the transport of gas in liquids and the subsequent reactions at triple-phase interfaces are poorly understood.
A joint research team led by Prof. Chen Jige of the Chinese Academy of Sciences’ Shanghai Advanced Research Institute (SARI) reported a real-time observation of the accelerated solid-liquid-gas etching progress of gold nanorods by introducing gas nanobubbles. They discovered that the underlying microscopic mechanism was affected by the thickness of the liquid layer.
Nature Materials published the findings.
The accelerated etching of gold nanorods with oxygen nanobubbles in aqueous hydrobromic acid can be observed in real time using liquid-cell transmission electron microscopy (TEM).
The researchers discovered that when an oxygen nanobubble was close to a nanorod below the critical distance (one nanometer), the local etching rate increased by more than one order of magnitude.
The strong attractive van der Waals interaction between the gold nanorod and oxygen molecules governed oxygen transport through the thin liquid layer, resulting in an increased etching rate, according to the results of molecular dynamics simulations.
This research sheds light on the rational design of solid-liquid-gas reactions for enhanced activities and offers a promising method for modifying the solid-liquid-gas reaction rate.