Carbon monoxide is an odorless and colorless gas made when fossil fuels burn incompletely. It’s also a silent killer. According to the Centers for Disease Control (CDC), more than 400 deaths and 20,000 emergency room visits are caused by carbon monoxide (CO) poisoning every year. While CO detectors and making sure your fireplace and heaters work correctly can help prevent exposure, treatment options are limited for those suffering from CO poisoning.
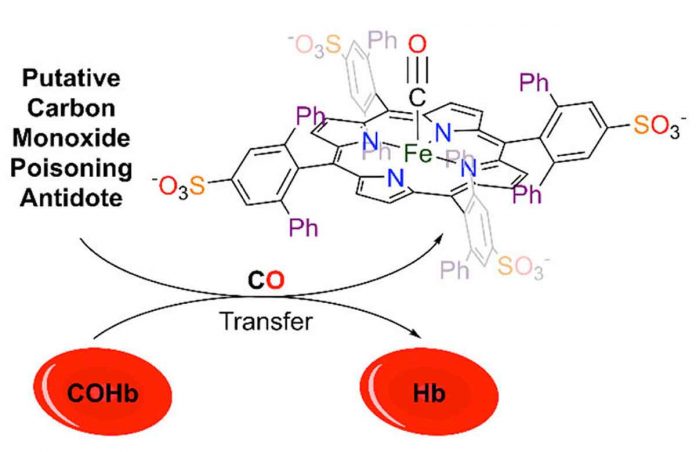
That’s why Tim Johnstone, an assistant professor of chemistry and biochemistry at UC Santa Cruz, has been working to develop an easy-to-administer antidote. Johnstone has been studying the chemistry of carbon monoxide. It is made of one oxygen atom and one carbon atom joined by a triple bond. In a biological context, CO binds to metal centers like the iron in hemoglobin. It then prevents this protein from functioning as it normally would, transporting oxygen from the lungs to tissues in the rest of the body.
Johnstone has designed small molecules that possess many of the features of the active site of hemoglobin but can bind CO much more tightly than the protein. His group described the ability of one such molecule to bind CO, sequester CO that is already bonded to hemoglobin, and rescue red blood cells exposed to CO, all promising signs for a future antidote.
Johnstone says these are early results. But the hope is to create a point-of-care treatment that can be administered quickly. The most common carbon monoxide poisoning symptoms are headache, dizziness, weakness, upset stomach, vomiting, chest pain, and confusion. Because it mimics the flu, people may experience symptoms without realizing the danger and delay seeking treatment.