Deutsches Elektronen-Synchrotron university scientists Andreas Stierle has found an alternative approach of storing hydrogen in a small nanoparticle made of a metal palladium. The palladium was only 1.2 nanometers in diameter.
This innovative method can transform nanoparticles into simple reservoirs to store hydrogen.
Hydrogen is regarded as an energy carrier for the future. This can provide climate-friendly fuels for airplanes and trucks. This also helps in producing climate-friendly steel and cement production and depends on the manner how hydrogen is produced.
But hydrogen storage is expensive. The gas needs to be kept at up to 700 bar or otherwise it needs to be liquefied. This means it will cool down to minus 253 degrees Celsius. Both the method needs additional energy.
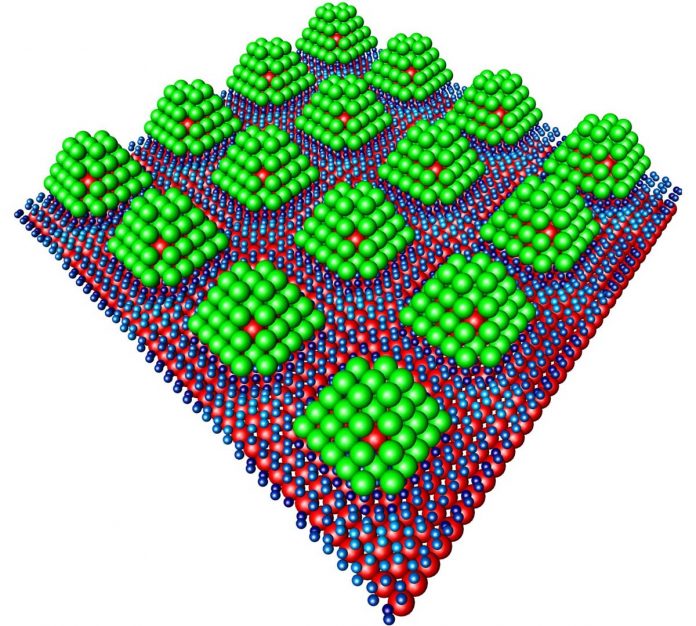
Palladium can absorb hydrogen like a sponge. This is why Stierle and his team has used palladium particles which are about the length of one nanometer.
To make sure that the palladiums are strong, the team has stabilized them by a core which is made of rare precious metal iridium.
Scientists reported that they can attach the palladium particles to the graphenes.
The scientists from Universities of Cologne and Hamburg have published a research paper of their findings in the ACS Nano.
DESY has utilized the X-ray source and observed what is happening when the palladium particles come into contact with hydrogen. They noticed the hydrogen is drowsing in the surfaces of the nanoparticles. Any hydrogen wasn’t penetrating inside the nanoparticles.
These nanoparticles are similar to chocolates. The chocolate has an “iridium nut” in the middle of it. The chocolate was covered in a palladium layer. On the outside of hydrogen, it was chocolate-coated.
We need stored hydrogen and a little heat. Hydrogen is emitted fast from the particles’ surface. Because the hydrogen does not need to push their way out from inside the cluster.
The scientists are looking for if storage densities can be achieved by this new method. Stierle said that there are still some problems in proceeding with real-world application of this method.
Scientists thinks other carbon structure forms can be a better carrier than graphene. Scientists are using carbon sponges that have small pores. Enough of palladium nanoparticles can fit inside these sponges.