Bats are extremely unusual animals. They are the only mammals with the ability to fly. Different species have adapted to feed on a wide range of foods ranging from mosquitos to fruit to blood. And, as investigations into the origin of the COVID-19 pandemic have revealed, they can harbour a plethora of viruses that are dangerous or fatal to other mammals without becoming ill themselves.
According to a study published in Science Advances, bats’ ability to survive as so-called viral reservoirs may be due in part to unique mutations. Such as the duplication of the gene encoding an antiviral protein known as protein kinase R. (PKR). According to the study, that second copy is the result of an ongoing evolutionary “arms race”. It has resulted in bats’ adaptation to and apparent immunity to a wide range of viruses over the course of their evolutionary history.
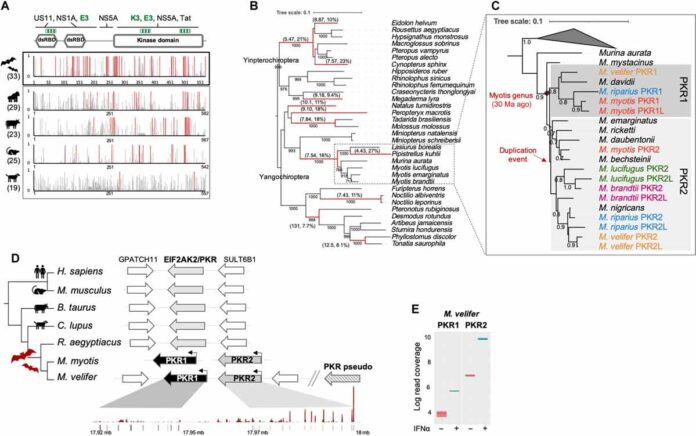
The researchers wanted to know how genetic similarities and differences between bats and other vertebrates affected their viral immunity. They specifically searched genomes for sequences encoding PKR. Study coauthor Stéphanie Jacquet, an evolutionary biologist at France’s Claude Bernard University Lyon 1, explains in an email that the team chose it for the comparison. Because it is conserved across invertebrates and important for immunity.
The researchers had to first sequence and assemble the genomes of 15 bat species. Then they focused on 33 of the more than 130 different species of mouse-eared bats (genus Myotis). As bat genomes are particularly scarce in the literature.
The researchers discovered that the gene EIF2AK2, which encodes PKR, rapidly evolved and underwent at least one duplication event early enough in bat evolution that the extra copy was present in every species they sampled. They discovered that some species had more than two copies of EIF2AK2. Or closely related sequences. Many of which encoded paralogs of PKR and shared its primary function as a frontline defence against viral invaders. These invaders blocks viral DNA and RNA translation. The team discovered that PKR duplication is unique to bats by comparing to those of humans, mice (Mus musculus) and dogs (Canis lupus familiaris).
The race against bat viruses
The researchers used gene editing to produce various bat PKRs or their orthologs,. Then exposed the cells to known kinase antagonists derived from bat-infecting viruses such as poxviruses, herpesviruses, and orthomyxoviruses. They discovered that PKR employs a variety of mechanisms to combat various viruses. It implied that viruses evolved to counteract bats’ existing defence mechanisms over time, and bats evolved new and improved PKRs in response. According to Alexa Sadier, an evolutionary developmental biologist at the University of California, Los Angeles who did not work on the study, this finding is a clear example of the Red Queen hypothesis. It is named after a character in Alice in Wonderland. And it posits that an evolutionary arms race occurs between predators and prey. Or in this case viruses and their hosts, in which the selective pressure imposed by an adaptation in one imposes new pressures.
What distinguishes bats?
The origins of the PKR duplication are unknown, as is why it did not occur in other mammals. One leading theory is that bats’ unusual immune abilities are related to the other distinguishing feature. It distinguishes them from the rest of their mammalian cousins: their ability to fly powered.
Understanding the mechanisms of host-virus interactions can lead to new strategies to prevent viral spillover from bats into other species.