University of Manchester scientists have created a molecular motor that consumes chiral fuel to drive rotation around a single covalent bond. The study was published in the journal Nature. Scientists described their work in developing a chemically powered directionally rotating motor and why they believe their efforts will result in similar systems being developed with other materials.
Previous research has shown that examples of biological motors exist in nature. ADT synthase is but one example. Such examples have served as inspiration for chemists attempting to replicate such motors and while some progress has been made. None had been developed that were able to rotate in a 360° axis around a single bond.
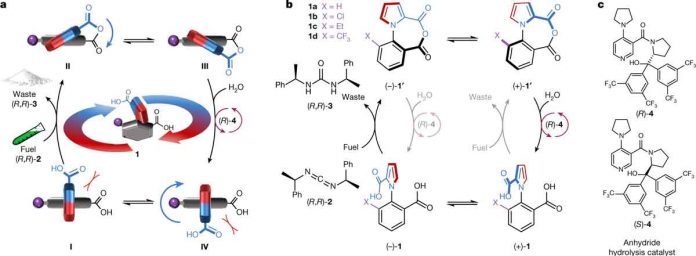
In the new study, scientists created a biological motor using 26 atoms. One where the motor was able to fully rotate around a single bond and continue rotating as long as it was fed fuel. Their motor consisted of two parts. A pyrrole-2 carbonyl group which made up the rotating part of the motor and a phenyl-2 carbonyl that served as a base. The two parts were connected together with a N-C bond. It also served as the axis for the motor. A carbodiimide fuel was used to drive the rotation. An intramolecular anhydride formed resulting in a hydrolysis reaction with a directional bias based on the chirality of the fuel. Another additive to speed up the process. Scientists noted that applying a diacid to the rotor and withdrawing fuel prevented the rotation when desired.
Testing showed the motor rotated at a rate of approximately one complete spin every three hours. They found it also spun in error. They suggest their work is just an initial foray into the creation of rotating motors. They expect future designs will rotate faster and that a means will be found to reduce the number of errors. Such motors could one day be used in applications such as mining metal ions from the ocean floor.